-
Projects
- Environment Box
- Passive Refrigeration
- Water Cooling
- Fog Catching
- Roof Geometries
- Optimal Insulation
- Cooler Windcatcher
- Green Machine
- Mitigating Humidity
- Convective Air Flow
- Styrene Reuse
- Thermal Reflection
- ETFE Rigidification
- Phase Change Materials
- Polar Reflection
- Cavity Depth Variation
- Vapor Permeability
- Algae Facade
- Moisture Buffering
- Engineered Geometries
- Recycled Desiccant Materials
- Living Wall
- Solar Shading Facades
- SHADESin.reACTION
- Low-Fab Dehumidification
- Breathing Wall
- Urban Heat Island
- Acoustical Design
- Latent Heat of PCM's
- Insulative Qualities of Air
- About
- Lectures
- Assignments
- Workshops
- Syllabus
- Resources
Results
The conventional air conditioning system driven by refrigerant vapor compression cycle is an inefficient process – more often than not overcooling air while condensing moisture from it. Decoupling the needs, cooling and dehumidifying, is an approach promising more energy efficiency. In hot and humid climates, especially those of least developed countries, a more energy efficient and cost effective way is imperative. The goal of this work is to prove that simple experimentation can be used as a tool to translate the inherent material qualities of low-cost, everyday materials into innovative building systems for air conditioning.
The first series of experiments test the same volume of material, a desiccant, to explore the materials performance without air flow. This is an effort to understand its most basic capacity for moisture absorption – condensing moisture from the air. The second series of experiments introduces air velocity to simulate air conditioning a space. This study asks how building materials can behave autonomously based on given environmental stimuli - humidity.
The first series of experiments test the same volume of material, a desiccant, to explore the materials performance without air flow. This is an effort to understand its most basic capacity for moisture absorption – condensing moisture from the air. The second series of experiments introduces air velocity to simulate air conditioning a space. This study asks how building materials can behave autonomously based on given environmental stimuli - humidity.
1.1 Series 1 - Materials & Procedures
This series uses a constant volume of desiccant to study the capacity for condensation over four hours. It monitors the ambient temperature and relative humidity. These observed values are then used to calculate the mass of water vapor in the controlled space.
Dehumidifying materials tested included a sponge, salt, silica gel, bentonite clay, and activated charcoal. These materials were selected based on their known adsorptive properties, affordability and accessibility.
Dehumidifying materials tested included a sponge, salt, silica gel, bentonite clay, and activated charcoal. These materials were selected based on their known adsorptive properties, affordability and accessibility.
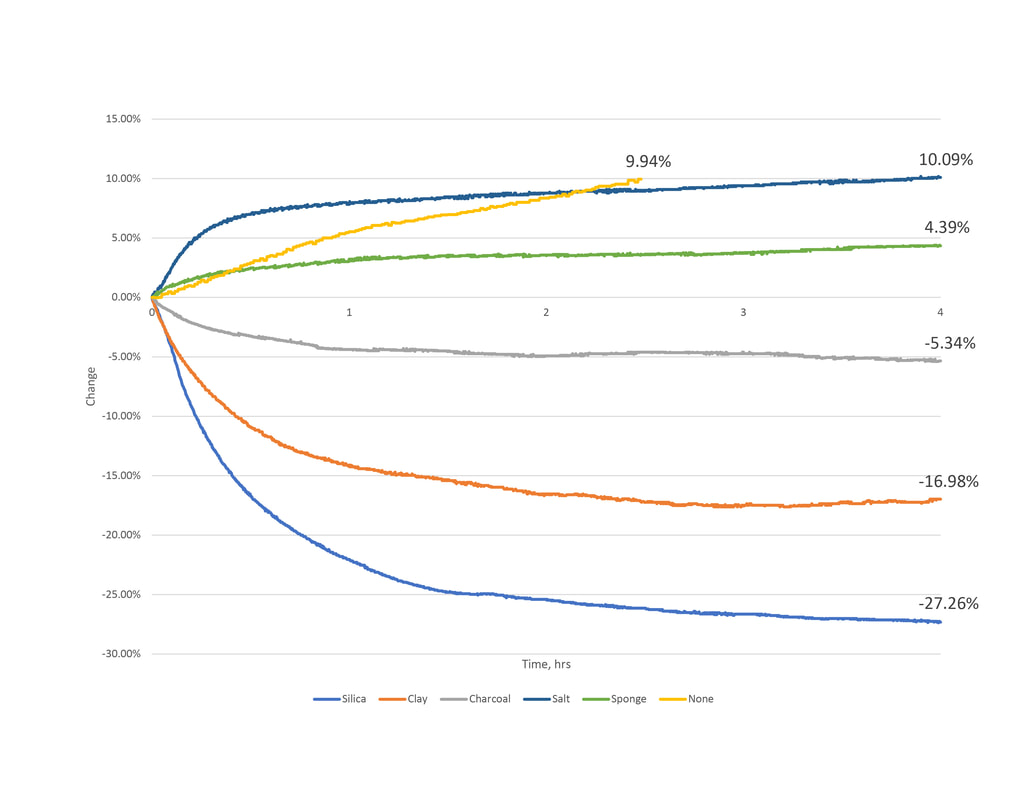
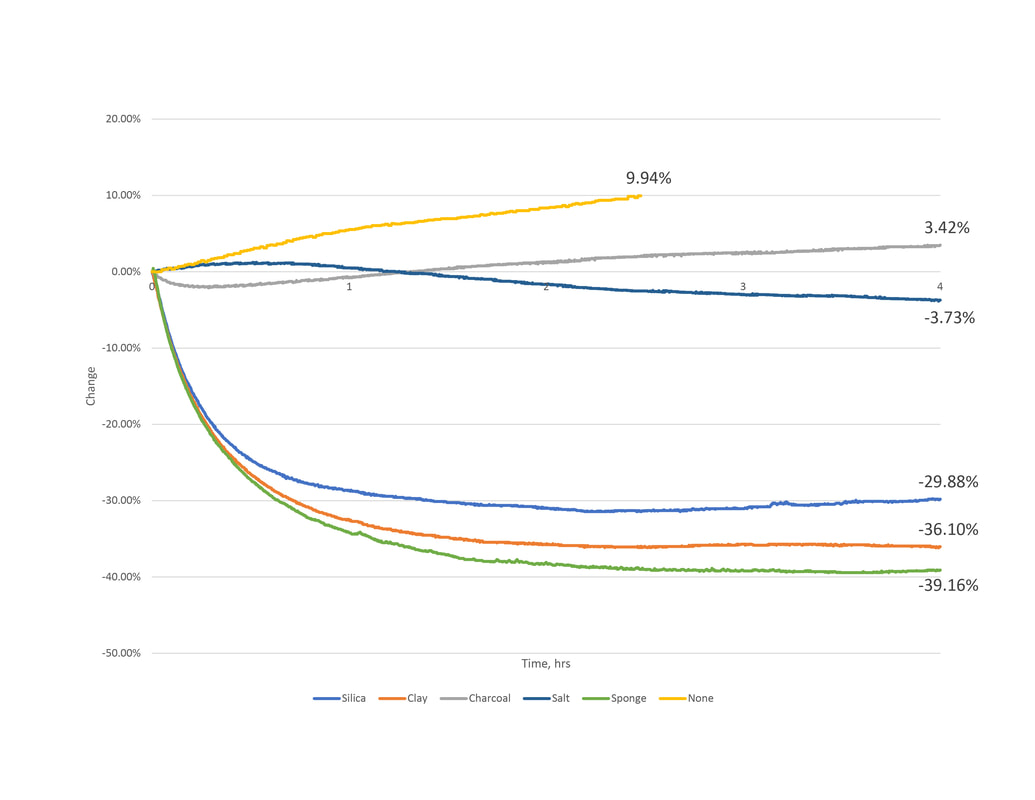
1.2 Series 1 - Results and Observations
None
A control test containing no material was conducted to compare to. Both test showed and steady increase of 5% per hour. Although this test was only conducted for two hours we can deduce that at 4 hours an increase between 15-20% could occur.
Charcoal
Activated charcoal tested well initially, having a negative change suggests that water content is being adsorbed. However the second test showed a positive change. Considering the rate of change is less than the test without material, it is understand that the charcoal is still doing work to slow humidification. Additionally the graphs change from negative to positive may mean the material's adsorption rate is best in only certain humidity conditions, thus performing poorly in other conditions.
Clay
The bentonite clay material had a two fold increase in capacity from test one to test two. This is due to a change in how we prepped and treated the material. Similar to silica gel packets, the clay purchased is packaged in packets too. The first test achieved a 17% reduction while remaining in this packaging. To understand if and how the packet packaging affects the material's capacity we removed the pellets from it in test two. This proved beneficial - the 36% reduction is the second highest performing material.
Rock Salt
Salt testing resulted in opposite manners from test one to test two. The first test showed a gain of water content in the environment box. The 10% increase is comparable to that when there is no desiccant material added. However, similar to the charcoals second testing, it's slower rate of change from the control test of no material suggests that it does indeed work - buffering humidification. The second test proves this. It yielded a 3% reduction of water content in the air.
Silica Gel
The silica gel packets had consistent results per testing of about 28% reduction of water vapor from the air. Similar to the clay we removed the gel beads from its packet packaging in test two. Unlike the clay, the silica's performance had minimal improvement.
Sponge
The sponge material testing resulted in contradictory manner between the two tests. In the first, the mass of water vapor in the air is increasing. An overall gain of .004 grams of water vapor mass was calculated – a 4.34% increase over time. We believe the sponge released moisture rather than condensing. The second, shows a difference of -.034 grams of water vapor mass – a 39% reduction over time. Although both were from the same packaging the noted difference is in the handling and preparation. For consumer marketing reasons new and unused sponges come moist out of air tight packaging. This saturated material was untreated prior to testing thus water moisture was released as it dried inside the controlled space. The second test accommodated this by preconditioning the testing material – ensuring that it was completely dry beforehand.
1.3 Series 2 - Materials & Procedures
This series introduces air flow. A fan is added move air across the desiccants tested. Ambient temperature and relative humidity are recorded for interior and exterior of the environment box. Just as previously, these observed values are then used to calculate the mass of water vapor in the controlled space. The materials tested here include silica gel, bentonite clay, and activated charcoal.
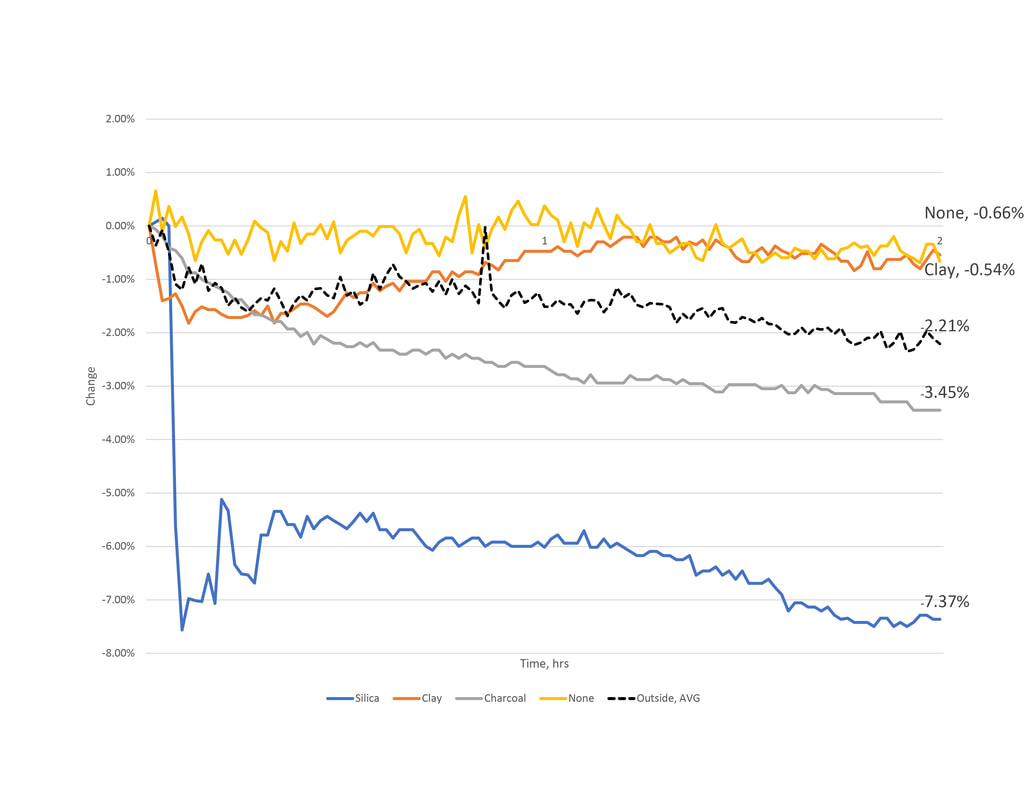
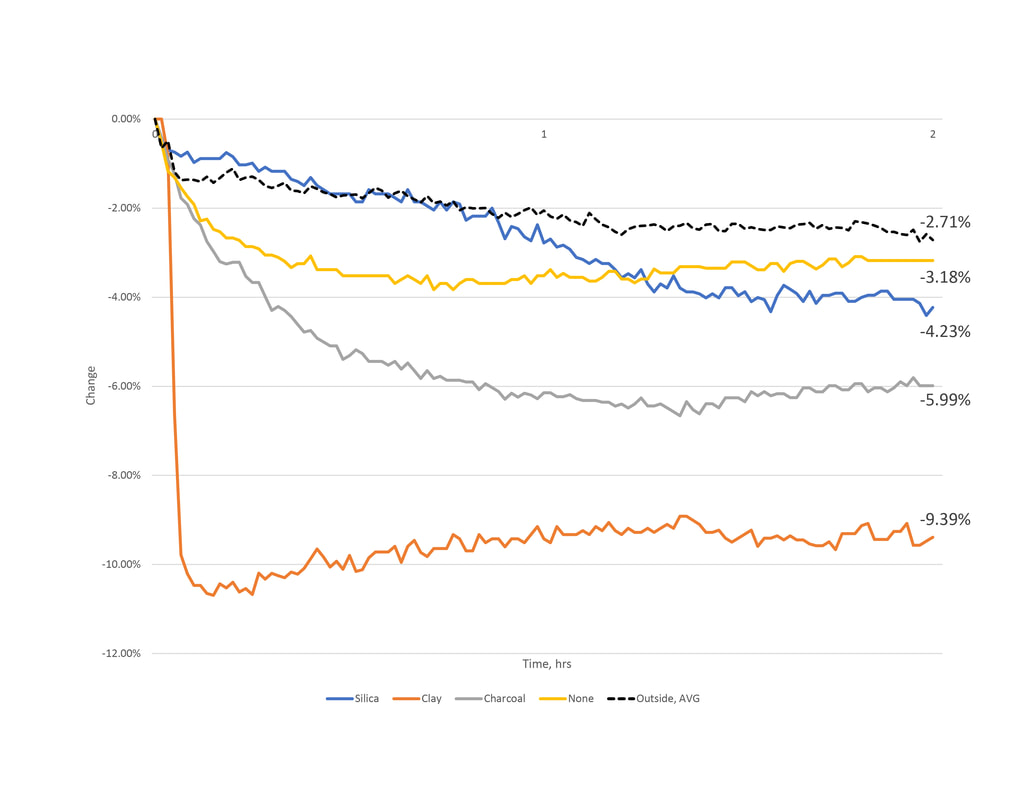
1.4 Series 2 - Results and Observations
Outside, Avg
During each of the subsequent material test, outside conditions were recorded. These have been averaged for a simplified understanding of how water content changed over time. Both calculations resulted around 2% reduction.
Charcoal
In series 1 the contradictory results of charcoal compelled us to test it again. Here, the results both showed a reduction of water content in the air and placed second best in performance. The second result almost twofold the first. As noted earlier, charcoal may perform better in specific conditions. Upon reviewing the collected data, the relative humidity during test 2 maintained within a range 5% less than test one. This could account for the change.
Clay
Unlike the consistent results in series 1, the clay's performance varies in series 2 experiments. The first test realized a 0.5% reduction after 2 hours. The second, close to a 10% reduction.
Silica
Similar to the first series of testing where the silica gel packets showed good capacity but little change between test, series two showed continued consistency. The interesting thing to note here is the relationship between rate and magnitude of change. In test one, the silica reached near 8% reduction in four hours. In the second testing conditions, the silica would only reach half of this in the same amount of time.
1.5 Conclusion
Each testing material had varying capacity - from buffering moisture change to adsorbing it. The hypothesis that silica gel material would impact moisture content inside the environment box due to its high capacity the greatest is partly true. Among each material, it has the most consistent capacity to adsorb water vapor from the air. This speaks to its efficiency and practicality as a potential dehumidifier - extending the thermal comfort.