-
Projects
- Environment Box
- Passive Refrigeration
- Water Cooling
- Fog Catching
- Roof Geometries
- Optimal Insulation
- Cooler Windcatcher
- Green Machine
- Mitigating Humidity
- Convective Air Flow
- Styrene Reuse
- Thermal Reflection
- ETFE Rigidification
- Phase Change Materials
- Polar Reflection
- Cavity Depth Variation
- Vapor Permeability
- Algae Facade
- Moisture Buffering
- Engineered Geometries
- Recycled Desiccant Materials
- Living Wall
- Solar Shading Facades
- SHADESin.reACTION
- Low-Fab Dehumidification
- Breathing Wall
- Urban Heat Island
- Acoustical Design
- Latent Heat of PCM's
- Insulative Qualities of Air
- About
- Lectures
- Assignments
- Workshops
- Syllabus
- Resources
Design Proposal
Abstract:
Urbanisation has lead to growth of humans. Migration to urban area from rural areas has crowded the cities. Also need to increase business has led to increase in high rise office buildings. It has become a trend to install curtain wall on almost office building. These glass facades traps heat inside; thus increasing the load on HVAC systems altogether. In hotter regions in summer it is really to achieve thermal comfort with air conditioning. Thus using solar shading facade might help reduce the heat due to radiation of sun. These facades would prevent direct sun rays from falling on the glass that would heat the interior. For this experiment I would consider Mumbai since it has hot and humid climate and is the financial capital of India, thus has more number of high rise office building.
Urbanisation has lead to growth of humans. Migration to urban area from rural areas has crowded the cities. Also need to increase business has led to increase in high rise office buildings. It has become a trend to install curtain wall on almost office building. These glass facades traps heat inside; thus increasing the load on HVAC systems altogether. In hotter regions in summer it is really to achieve thermal comfort with air conditioning. Thus using solar shading facade might help reduce the heat due to radiation of sun. These facades would prevent direct sun rays from falling on the glass that would heat the interior. For this experiment I would consider Mumbai since it has hot and humid climate and is the financial capital of India, thus has more number of high rise office building.
Research Questions:
•Shading Devices prevents sun radiation falls directly on the glass.
•That reduces heating of internal spaces.
•Thus, it will reduce the demand for cooling system.
•We have to know how much shading devices in this case louvres help reduce the internal temperature by shading the shading glass?
•That reduces heating of internal spaces.
•Thus, it will reduce the demand for cooling system.
•We have to know how much shading devices in this case louvres help reduce the internal temperature by shading the shading glass?
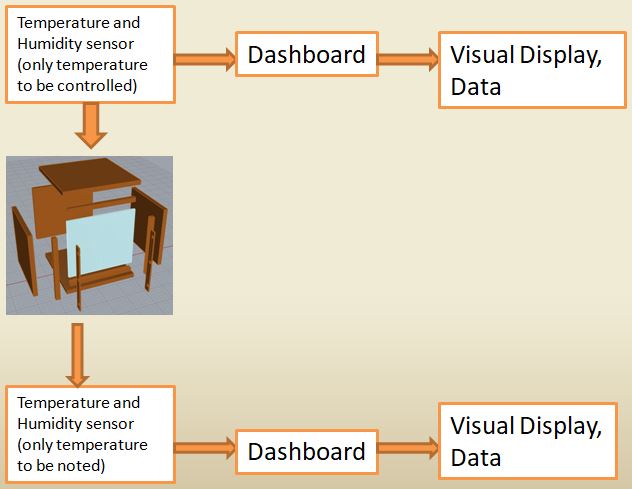
Mockup
- This is a 1 1/2 ft x 1 1/2 ft x 1 1/2 ft wooden box with 3 solid side.
- One side comprising of acrylic sheet.
- The piece with the slits for louvres is attached to the frame of the glass.
- The slits for louvres have angles 0 and 30.
- The lamp should be placed at certain angle to represent sun.
- The temperature from inside and outside the box before and after put on the panels is to be noted.
Electronic Control:
Hypothesis:
Glass traps heat inside the building. Shading the glass with louvres will help reduce the greenhouse effect by avoiding or reducing the direct radiation from the sun.
Objectives:
To avoid or reduce the solar radiation on the curtain glass to minimise the green house effect that is caused it the building.
To design the assembly of louvres to help control radiation on curtain glass.
To reduce the load on HVAC system by achieving thermal comfort with passive strategies.
To design the assembly of louvres to help control radiation on curtain glass.
To reduce the load on HVAC system by achieving thermal comfort with passive strategies.
Schedule
Week |
2/24-2/27 |
3/8-3/14 |
3/15-3/21 |
3/22-3/28 |
3/29-4/4 |
4/5-4/11 |
4/12-4/18 |
4/19-4/25 |
4/26-5/2 |
5/2-5/9 |
Work |
Mock up |
Build the final experiment box |
Build the final experiment box |
Without Louvres |
Louvres at 0 |
Louvres at 30 |
Data Sorting and Review |
Work on mistakes |
Work on mistakes |
Final Presentation |
Materials Required
|
For box
Plywood: $20
Acrylic: $10 Temperature and Humidity Sensor: Elego Halogen Lamps: IDRL Pink Insulation Foam: $49.83 (for 1/4" x 4' x 50') Stand for lamp: |
Electronics
2 Temperature sensors: $2.02 (each)
Arduino Uno R3 with USB cable: $9.99 Breadboard: $5.54 |